A novel type of contractile ring enables cell division
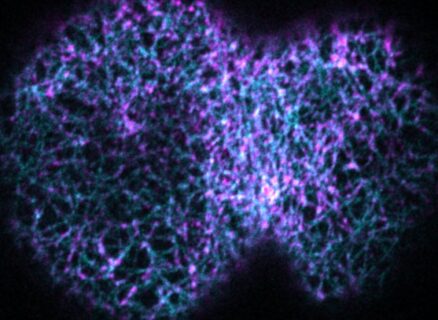
During cell division the cellular content of the mother cell must be equally segregated between the two daughter cells. In animal cells, cell division is mediated by a ring-like apparatus consisting of long filamentous actin fibers. The actin ring assembles around the cell equator and shrinks in size with the help of the motor protein myosin II. Actin ring constriction results in cleavage furrow formation and the generation of two new daughter cells. In case the formation of the actin fibers is defective, no contractile ring forms and the division of the mother cell fails. Cell division failure can facilitate the development of numerous malignancies including infertility and cancer. Surprisingly, a previous study had shown that cell division can still succeed even when actin fiber assembly is defective, in case another component of the ring-like apparatus is removed. How cell division and cleavage furrow formation is accomplished in this case was a mystery.
The research group of Prof. Dr. Esther Zanin answered this question now using advanced live-cell imaging in the small nematode C. elegans. They discovered that in this situation another protein called anillin forms linear fibers, which assemble into a ring-like structure at the cell equator. Subsequently, linear anillin fibers and the motor protein myosin II mediate cleavage furrow formation and cell division. To understand how anillin might form linear fibers and induce cleavage furrow formation, the authors deleted different regions of the anillin protein and analyzed the phenotypic consequences. The mutational analysis of anillin uncovered that an uncharacterized and unstructured region was essential for forming linear fibers and cleavage furrow formation. Such unstructured regions lack a distinct three-dimensional structure and are instead highly flexible and dynamic. “How this dynamic region of anillin forms linear fibers that are able to shorten in length and finally mediate cleavage furrow formation are exciting questions to pursue in future studies” explains Esther Zanin.
Anillin is not only present in C. elegans but also in humans and even the unstructured anillin region exists in the human protein. Point mutations in the unstructured anillin region can cause kidney failure and therefore, the principles discovered in the small nematode might also have implications for human health and disease.
Originalpublikation:
Anillin forms linear structures and facilitates furrow ingression after septin and formin
CellReports, 42 (2023) 113076. doi:10.1016/j.celrep.2023.113076
Prof. Dr. Esther Zanin
Department Biologie
Professur für Experimentelle Molekulare Zelldynamik (Prof. Dr. Zanin)
- Phone number: +49 9131 85-25056
- Email: esther.zanin@fau.de